Avda. Francisco Vallés, 8 47151
Boecillo (Valladolid) - Spain
(+34) 983 54 98 96
- e4Risk
- Why ApoE4?
- Key Features
- Technical Information
- Submit a budget request
- Submit a purchase order

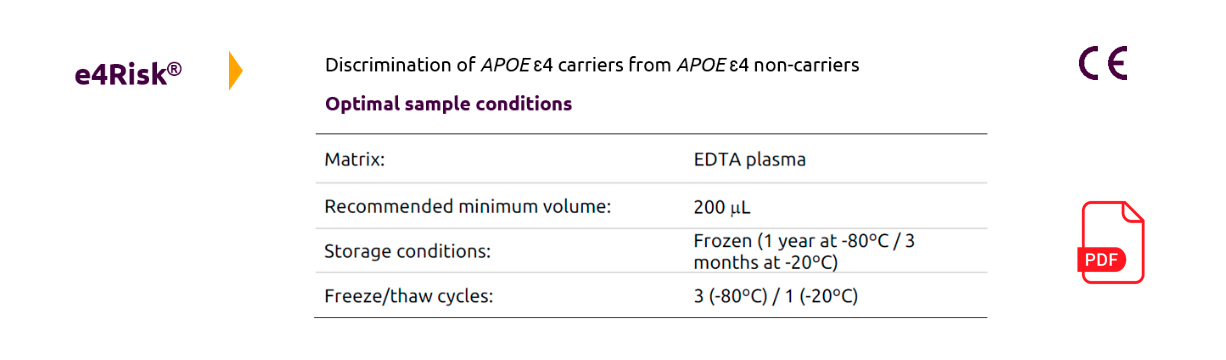
The e4Risk ® is a non-genetic, cost-effective, and highly reliable method to detect the presence of the ApoE4 protein in human blood plasma using high-throughput chemistry analyzers.
This is achieved using a turbidimetry-based assay with excellent performance in the context of the hospital/clinical settings (100% sensitivity and 99% specificity).
CE Marking. The process of obtaining the FDA approval for the e4Risk® is currently ongoing.
*Test based on patent nº EP15382537.7: “Methods for apolipoprotein detection”.

Apolipoprotein E is a glycoprotein involved in lipid metabolism. It is encoded by the APOE gene, which has three different alleles (ε2, ε3 and ε4) that encode three different ApoE isoforms (ApoE2, ApoE3 and ApoE4, respectively). The allele ε4 of APOE gene is the major genetic risk factor and the second most important risk factor after age for non-dominantly inherited AD (by far, the most common form). APOE ε4 is present in approximately 20% of the global population and in 40-60% of all patients with AD2,3.
The presence of one allele ε4 of the APOE gene increases the risk of AD by 3-5 fold, while the presence in homozygosis increases the risk by 8-12 fold4. Furthermore, APOE ε4 carriers have an earlier onset of the disease5 and they progress faster from the prodromal stage of AD (mild cognitive impairment, MCI) to AD than APOE ε4 non-carriers6. Therefore, the identification of APOE ε4 carriers can clearly benefit clinical practice, clinical trials and prevention strategies.
Clínical Practice
Brain amyloidosis (extracellular amyloid beta (Aβ) accumulation within the cerebral tissue) is considered one of the earliest events in AD pathophysiology and a key factor for AD diagnosis according to recent guidelines1. As a matter of fact, it was recently reported that brain amyloidosis is considered a determinant information not only for AD diagnosis but also for patient management7.
Brain amyloidosis can be determined accurately through measurement of Aβ concentration in the cerebrospinal fluid (CSF) or with imaging techniques (amyloid positron emission tomography (amyloid-PET)). However, these techniques are invasive and/or expensive and it is not possible to prescribe these tests to all patients.
Additionally, they require specialized instrumentation and personnel and they are not available in all hospitals. There is an actual need of non-invasive and cost-effective test that can be easily implemented in the clinical practice for selection of patients to be subjected to CSF analysis or amyloid -PET for brain amyloidosis evaluation.
There are strong evidences that show that ApoE4 is closely related with brain amyloidosis. APOE ε4 carriers have twice as much probability of having a positive result on brain amyloidosis in the prodromal stage of the disease (mild cognitive impairment) 8 when compared to APOE ε4 non-carriers, and this probability increases up to 90-97% in the symptomatic phase of AD9. Therefore, ApoE4 is a reliable predictor of brain amyloidosis, especially in patients with symptoms of dementia.
e4Risk® is very reliable and cost-effective test that can detect APOE ε4 carriers with a blood sample and using the already available instrumentation of clinical laboratories and hospitals. These features make e4Risk® an optimal solution for detection of APOE ε4 carriers, and therefore of patients at high risk of brain amyloidosis, within the clinical practice. e4Risk® will provide a very relevant information that will help the clinician, along with the age and the results of other routine tests, to select patients for additional tests to confirm brain amyloidosis and help in the AD diagnosis process.
Likewise, a positive result in e4Risk® could help to determine the presence of brain amyloidosis in patients with mild cognitive impairment that are not candidates for additional tests for brain amyloidosis, since approximately 80% of APOE ε4 carriers older than 80 years with mild cognitive impairment have brain amyloidosis8.
The inclusion of e4Risk® in the initial routine test panel for cognitive impairment and dementia would accelerate the obtention of AD diagnosis and reduce the uncertainty time before diagnosis, since it would help clinicians to select specific confirmatory tests for the disease. In the mid-term, this test might be used in Primary care, where it could be incorporated into the clinical criteria for derivation of patients with subjective memory complaints to the specialist.
Clínical Trials
Currently, AD research is focused in early stages of the disease, where most of clinical trials are designed to include people at-risk. e4Risk®, due to its ease of use and low cost can be very useful as a screening test for AD clinical trials, where APOE ε4 carriers can be selected in a fast and inexpensive manner.
Prevention
Prevention has become a major objective in the AD Health strategy. The failure during the last two decades of all investigational drugs led to the idea that treatments should be directed to very early stages of the disease10,12.Identification of people at risk is key for the effectiveness of preventative measures and consequently, APOE ε4 carriers were defined as a priority population for prevention13,14.
Key features

Technical information
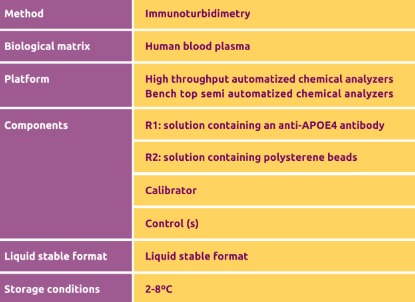
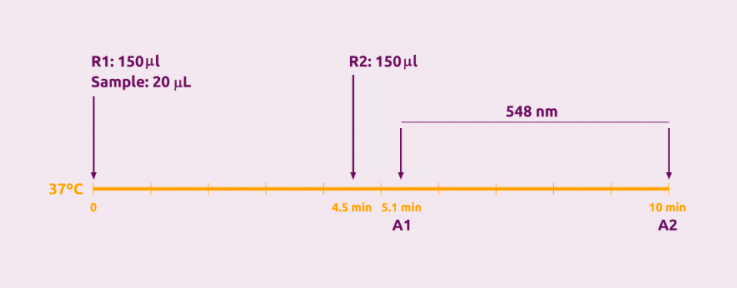
Assay Methodn
| Reference | Name | R1 | R2 | CAL | C+ | C- | Insert |
|---|---|---|---|---|---|---|---|
| 3135015 | e4Risk® kit | 1 x 10 ml | 1 x 10 ml | 1 x 0.5 ml | 1 x 0.5 ml | 1 x 0.5 ml | |
| 3135005 | e4Risk® Reagents | 1 x 20 ml | 1 x 20 ml | 1 x 1 ml | 1 x 1 ml | 1 x 1 ml | |
| 3135010 | e4Risk® Reagents | 2 x 10 ml | 2 x 10 ml | ||||
| 3935005 | e4Risk® Calibrator | 2 x 0.5 ml | |||||
| 3935010 | e4Risk® Calibrator | 4 x 0.5 ml | |||||
| 3935020 | e4Risk® Controls | 1 x 0.5 ml | 1 x 0.5 ml |




